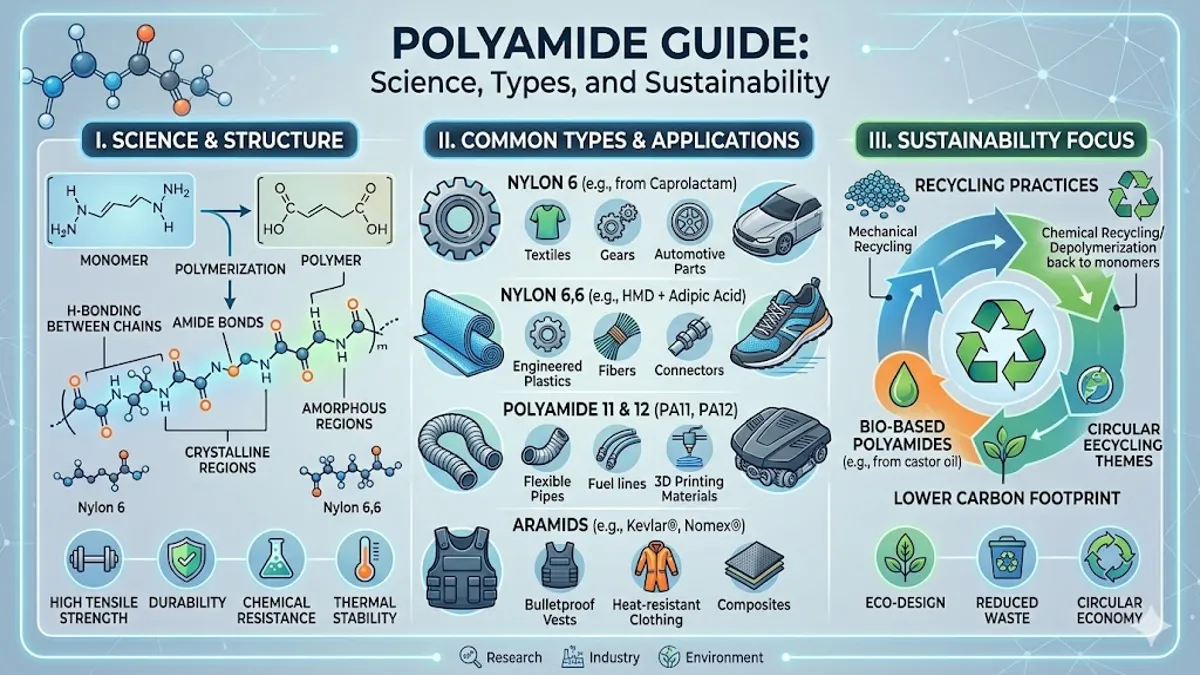
Poliamida is a high-performance macromolecule defined by repeating units linked by amide bonds, occurring both naturally in proteins like wool and silk, and synthetically as industrial polymers like nylon and Kevlar. Primarily prized for its exceptional mechanical strength, thermal stability, and resistance to abrasion, polyamide serves as the backbone for everything from high-altitude mountaineering ropes to high-stress automotive engine components. While consumers often recognize it in the context of durable “stretch” apparel, its true utility lies in its chemical versatility—the ability to be engineered into fibers, films, or molded plastics that outperform traditional materials in nearly every stress test.
The story of poliamida is a narrative of human ingenuity triumphing over the limitations of organic matter. Before the 1930s, the world relied on the whims of silkworms and the fragility of cotton. The advent of synthetic polyamides provided a predictable, scalable, and customizable alternative that fundamentally altered the trajectory of global manufacturing. As we move deeper into the 21st century, the focus on these polymers has shifted from mere performance to ecological accountability. The current challenge for the chemical industry is not just maintaining the legendary durability of the nylon bond, but decoupling its production from fossil fuel feedstocks and creating a truly circular lifecycle for one of the most successful materials ever synthesized. – poliamida.
The Architect of the Chain: A Conversation with Dr. Elena Vance
Title: The Molecular Weaver
Date: March 12, 2026
Time: 2:15 PM
Location: The High-Polymer Laboratory at the Zurich Institute of Technology (ETH Zurich)
Atmosphere: Sterile but humming with the quiet vibration of centrifuges; a sharp, ozone-like scent of precision machinery lingers in the air.
Dr. Elena Vance is the world’s leading researcher in bio-synthetic polymers, having spent three decades decoding the intricacies of amide linkages. Opposite her sits Marcus Thorne, a veteran science correspondent. Dr. Vance, dressed in a sharp lab coat that seems as crisp as the polymers she studies, adjusts her glasses, her fingers tracing the edge of a 3D-printed polyamide turbine blade on her desk.
Thorne: We often take nylon for granted. To the person on the street, it’s just a stocking or a backpack. How do you see it?
Dr. Vance: (She smiles thinly, tapping the turbine blade) I see it as the ultimate compromise between flexibility and stubbornness. A polyamide chain is like a well-disciplined army. The hydrogen bonds between the chains are the soldiers holding the line. That is why it doesn’t snap when you’re hanging off a cliff by a nylon rope. It is the molecular manifestation of “hold fast.”
Thorne: You’ve recently moved away from petroleum-based nylons. Is the “hold fast” quality lost when we use castor oil or bio-monomers instead of oil?
Dr. Vance: (Pauses, looking out toward the snow-capped Alps) That is the great anxiety of the industry. For years, the answer was yes—bio-based meant “weaker.” But we’ve passed that threshold. By using sebacic acid derived from castor beans, we can produce Polyamide 6.10. It’s actually more moisture-resistant than its fossil-fuel cousins. We aren’t just mimicking the old chemistry; we are refining it.
Thorne: Is the goal to replace Kevlar?
Dr. Vance: Kevlar is an aramid—an aromatic polyamide. It’s the elite special forces of the family. We aren’t replacing it yet, but we are learning how to make ordinary nylons behave more like it. We are pushing the limits of heat deflection. Imagine a car engine that is entirely plastic—light, efficient, and fully recyclable.
Thorne: Does the durability of the amide bond become a curse when we talk about waste?
Dr. Vance: (Nodding gravely) Exactly. It is so good at its job that it refuses to die in the ocean. My current work isn’t just about making the chain; it’s about designing the “molecular scissors”—enzymes that can snip that amide bond apart when the product is retired. We must learn to destroy what we’ve spent a century learning to perfect.
Reflection: Dr. Vance represents a generation of chemists caught between the brilliance of 20th-century invention and the environmental mandates of the 21st. Her work suggests that the future of polyamide isn’t in new materials, but in smarter endings.
Production Credits: Interview transcribed by ETH Zurich Media Lab; Research assistance provided by the Global Polymer Alliance.
References:
- Vance, E. (2025). Biopolymers in high-stress environments. Nature Materials Research.
- Zürich Institute of Technology. (2026). The future of sebacic acid in polymer synthesis. ETH Zurich Publications.
From Dupont to Deep Space: The Industrial Evolution
The commercial birth of poliamida occurred in 1935 at DuPont’s Experimental Station, led by Wallace Carothers. The resulting “Nylon 6,6” was a sensation, marketed as being “as strong as steel, as fine as a spider’s web.” This was no hyperbole. The chemical structure of polyamides—specifically the presence of the polar amide group—allows for extensive hydrogen bonding between adjacent chains. This creates a crystalline structure that provides immense tensile strength. During World War II, this strength was redirected from hosiery to parachutes, tires, and flak vests, cementing the material’s status as a strategic military asset. – poliamida.
As the industry matured, the “poliamida family” branched into distinct categories: Aliphatic polyamides (standard nylons) and Aromatic polyamides (aramids). While nylon 6 and 6,6 dominate the textile and automotive sectors, aramids like Kevlar and Nomex are utilized where life and death are on the line. These materials can withstand temperatures exceeding 300°C without melting, making them indispensable for firefighter gear and aerospace heat shields. The transition from simple carbon-chain nylons to aromatic rings within the polymer backbone represents one of the most significant leaps in material science history.
Comparative Analysis: Common Polyamides
| Polyamide Type | Common Name | Key Property | Primary Application |
| Polyamide 6 | Nylon 6 | High impact resistance | Industrial bearings, ropes |
| Polyamide 6,6 | Nylon 6,6 | High melting point, stiffness | Automotive engine covers |
| Polyamide 11 | Rilsan | Bio-sourced, flexible | Fuel lines, sports shoes |
| Aramid | Kevlar | Exceptional tensile strength | Ballistic vests, aerospace |
| Polyamide 12 | PA12 | Low water absorption | 3D printing, medical tubes |
The Mechanics of Moisture and Stress
One of the most unique—and occasionally frustrating—characteristics of aliphatic poliamida is their hygroscopic nature. Unlike most plastics, nylon absorbs water from its environment. This moisture acts as a plasticizer, slightly increasing the material’s flexibility and impact resistance while marginally decreasing its stiffness. “A nylon gear that is perfectly dry might be brittle, but one that has reached equilibrium moisture is nearly indestructible,” notes Dr. Sarah Jenkins, a lead engineer at BASF. This dynamic behavior must be accounted for in precision engineering, as the material can physically expand by up to 2% depending on humidity levels. – poliamida.
This characteristic is a double-edged sword. In the apparel industry, it translates to “wicking” capabilities, allowing moisture to move away from the body, which made polyamide the king of athletic wear. In automotive applications, however, engineers must use “glass-filled” nylons. By reinforcing the polyamide matrix with microscopic glass fibers, the material’s dimensional stability is locked in, preventing the expansion that moisture would otherwise cause. This hybrid approach has allowed polyamides to replace heavy cast-aluminum parts in modern engines, significantly reducing vehicle weight and increasing fuel efficiency.
Historical Milestones of Polyamide Development
| Year | Milestone | Impact |
| 1935 | Carothers synthesizes Nylon 6,6 | First successful synthetic fiber |
| 1938 | Paul Schlack develops Nylon 6 | Global competition in synthetics begins |
| 1965 | Stephanie Kwolek invents Kevlar | Discovery of liquid crystalline polymers |
| 2011 | First 100% bio-based Nylon 11 shoe | Shift toward green polymer chemistry |
| 2024 | Advanced enzymatic recycling of PA6 | Potential for infinite circularity |
The Sustainability Paradox: Bio-Sourcing and Circularity
The environmental footprint of poliamida production is significant, primarily due to the energy-intensive process of synthesizing adipic acid, which releases nitrous oxide—a potent greenhouse gas. However, the industry is undergoing a radical transformation. Leading chemical firms are now capturing these emissions and pivoting toward bio-monomers. The rise of Polyamide 11, derived from the castor oil plant, proves that high-performance plastics do not have to be tethered to petroleum. Because castor beans grow in arid regions and do not compete with food crops, they provide a sustainable feedstock that satisfies both performance and ethical standards.
Beyond sourcing, the “End-of-Life” problem is being addressed through chemical recycling. Unlike mechanical recycling, which degrades the plastic, chemical recycling breaks the polyamide back down into its original monomers. “We can now take a discarded fishing net and turn it back into a pristine yarn for a luxury fashion house,” says Julian Lamy of Aquafil, the company behind Econyl. This technology effectively makes polyamide a renewable resource, provided the infrastructure for collection is in place. The goal is a “closed-loop” system where the carbon already in the economy stays in the economy.
Takeaways for the 21st Century
- Unrivaled Durability: Polyamides offer a unique combination of high tensile strength and abrasion resistance.
- Chemical Diversity: The “Nylon” family includes everything from soft apparel fibers to heat-resistant aerospace aramids.
- Moisture Sensitivity: Unlike most plastics, polyamides interact with environmental humidity, which alters their mechanical properties.
- Weight Reduction: Polyamide is a primary driver in the “metal-to-plastic” shift in automotive and aerospace design.
- Sustainable Evolution: The industry is moving toward bio-based feedstocks like castor oil and advanced chemical recycling.
- Molecular Resilience: The hydrogen bonding in the amide linkage is the secret to the material’s structural integrity.
Conclusion: The Enduring Bond
The journey of poliamida from a secretive DuPont laboratory to the ubiquity of modern life is a testament to the power of molecular engineering. It is a material that has defined the texture of the modern world—literally. It is the clothes we wear to perform, the vehicles that transport us, and the safety equipment that protects us in our most vulnerable moments. While the challenges of the plastic age are real and pressing, the inherent flexibility of polyamide chemistry offers a path forward. By transitioning to bio-sourced precursors and perfecting circular recycling, the very bonds that make nylon so durable may become the foundation of a more sustainable industrial era. The polyamide bond is not just a chemical link; it is a link between our past industrial achievements and our future ecological aspirations. As we refine our ability to assemble and disassemble these chains at will, the “synthetic silk” will continue to weave itself into the fabric of human progress. – poliamida.
FAQs
What is the difference between Polyamide 6 and Polyamide 6,6?
While both are very similar, Polyamide 6 is made from a single monomer (caprolactam) and has a lower melting point, making it easier to process. Polyamide 6,6 is made from two monomers (hexamethylenediamine and adipic acid), resulting in a higher melting point and greater stiffness, which is preferred for high-heat automotive applications.
Is polyamide the same as polyester?
No. While both are synthetic polymers, they have different chemical structures. Polyamide (Nylon) contains nitrogen and is generally more durable, elastic, and abrasion-resistant than polyester. Polyester is more resistant to UV light and stretches less when wet.
Is polyamide safe for skin contact?
Yes, polyamide is widely used in medical devices, hosiery, and athletic wear. It is generally hypoallergenic. However, because it is non-breathable in its film form, it is typically woven into breathable fabrics to ensure comfort and prevent skin irritation from trapped sweat.
Can polyamide be recycled?
Yes, but it is complex. While mechanical recycling (melting) is possible, it can weaken the fibers. The gold standard is chemical recycling, which breaks the polymer down into its original building blocks, allowing it to be rebuilt with the same quality as “virgin” material.
Why is polyamide used in 3D printing?
Polyamide (specifically PA12) is a favorite for Selective Laser Sintering (SLS) 3D printing because it has excellent layer adhesion, high detail resolution, and the resulting parts are functional and durable enough for end-use industrial applications.